
CBSE Class 10 Sample Paper
Exams are on the head and students are looking for the best study material to score good marks in their exam. With a lot of books and study materials, it becomes very difficult to decide what to read and what not to read. So, today we are sharing with you CBSE Class Sample Paper for Class 10. You can also download the Class 10 CBSE Sample Paper PDF below.
CBSE Sample Paper: Class 10
Q.1. How is the concentration of hydronium Ions affected when a solution of an acid is diluted?
The concentration of hydronium Ion will decrease when an acid solution is diluted with water
Q.2. In the following food chain, 100 J of energy is available to the Lion how much energy was available to the producer
Plants >>deer >>lion
Q.3. State two advantages of conserving
Forest and wildlife
Answer 3.
Conserving forests help us to retain the subsoil water it also helps to maintain biodiversity of living things
Floods can be prevented from this.
Q.4. State Two ways for preventing rusting of iron articles.
We can use the galvanization process in which we apply a layer of zinc coating over the iron object so that they do not come in contact with the oxygen and moisture. We can apply Paint on the iron object so as to reduce their contact with moisture and oxygen
Question 5 list the properties of magnetic lines of force
- The magnetic field lines of a magnet will always form of continuous closed path loop.
- The magnetic lines of a magnet starts from the north pole and ends in to the South Pole
- You will never see these magnetic lines intersecting with each other
Question 6 Differentiate between Metals and nonmetals on the basis of their chemical properties
When we react a metal with dilute acid they will displace the hydrogen present in the acid
Displacement can be easily noticed in the case of metals
Basic oxides are formed on the metals react with oxygen present in the air
Nonmetals
Non-metals from the acidic oxides when they react with oxygen is present in the air
The displacement which we see in the case of metals cannot be observed in the case of non-metals and as they reacted with dilute acids
Non-metals do not show the displacement reaction like in the case of metals
Question 7: Explain the process of nutrition in amoeba
Amoeba is a unicellular organism and it uses its finger-like projection which is called as pseudopodia. it forms of food vehicle inside its body where the complex food gets broken into simpler substances for digestion. enzymes help them to break into simpler substances which then get diffuse into the cell cytoplasm. the food which remains undigested is thrown out of the amoeba by the process of endocytosis.
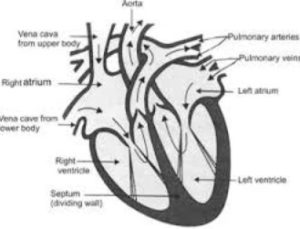
Q.8. Draw labelled diagram of the human heart
Question 8: What is reflex arc draw labelled diagram to show reflex arc on touching a very hot object
Reflex action is a spontaneous action which can be noticed in our body when we face an instant stimuli. The complete path in which the reflex action takes place is known as the reflex arc
Question 9. Show how you would connect three resistors each of 6 ohm so that the combination has a resistance of
(a) 9 ohms
(b) 4 ohms
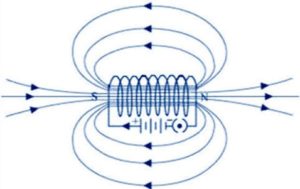
Question 10
Draw sketch of the pattern of field lines due to a:
(a) Current flowing into a circular coil
(b) solenoid carrying current
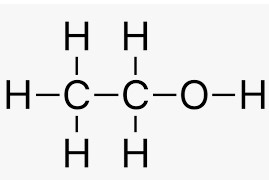
Question 11 Write the structural formula of Ethanol. What happens if it is heated with an excess of concentrated sulphuric acid and 443 K?
Question 12: Write the chemical equation for the reaction stating the role of concentrated H2 S o4 acid in this reaction
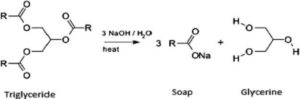
Question 13. Distinguish between esterification and saponification reaction with the help of chemical equation for each. State one use of each esters and saponification process.
Esterification: The reaction in which are carboxylic acid combines with and alcohol and forms an Ester are known as esterification
In esterification, esters are formed
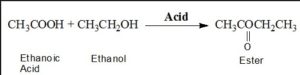
Saponification: Esters react under the presence of acid or base to give back alcohol and carboxylic acid.
Alcohol is formed
Latest posts by Deepak Kumar (see all)
- DAV Class 7 Science Solutions With Explanations: All Chapters - February 6, 2022
- Pollution of Water Question Answers | DAV Class 8 Science - February 4, 2022
- Fabric from Fibre Solutions | DAV Class7 Science Chapter 14 - January 25, 2022

Leave a Reply